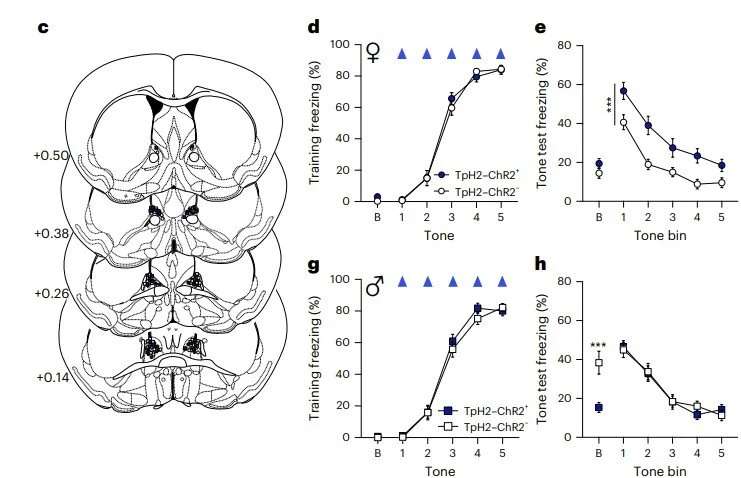
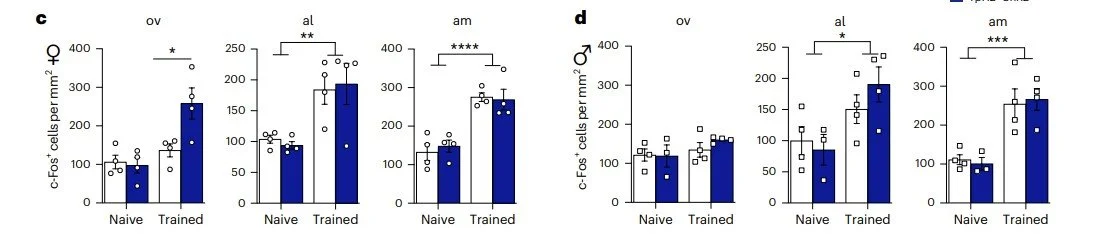
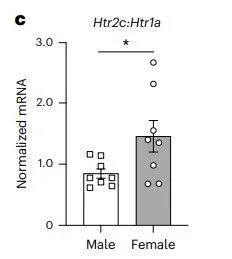
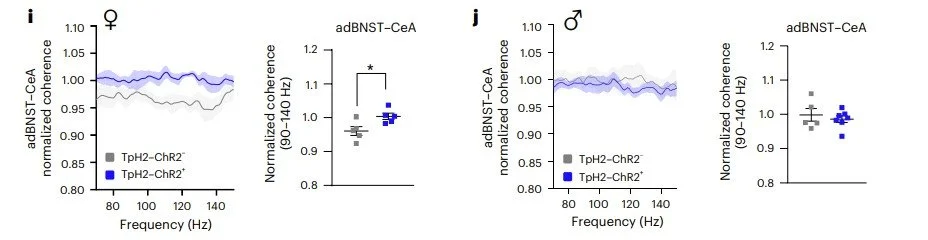
Mia won the Stanley Weiss Award in Experimental Psychology at the Eastern Psychological Association Conference and a Travel Award at the The Graduate Center Psychology in Behavioral Cognitive Neuroscience Program.
Mia at the 2026 EPA Conference
Tikva was awarded the Doctoral Student Research Grant from the Biology Program in Neuroscience at The Graduate Center.
Tikva at The Graduate Center, CUNY
Thanks to these awards, watch for their work on the physiology and function of fear extinction networks at upcoming conferences!